

Dermalize PHANTOM ULTRA-THIN FILM ROLL (STERILE) INNOVATION 2023 – STERILE
Dermalize Phantom – Ultra-thin protective tattoo film is an innovative product designed to give tattoo collectors the highest level of comfort. It is half of the regular thickness (0,01mm) of most of similar products available on the market; it is extremely flexible and comfortable to wear, sterile and its matt finish makes it almost invisible on the tattooed skin.
Dermalize Phantom – Ultra-thin protective tattoo film is available for professional tattoo artists in roll format (15cm x 10mt), ideal for the first application after finishing the tattoo.
Dermalize Phantom – Ultra-thin protective tattoo film is a latex-free breathable film, made of sterile ultra-thin polyurethane and hypoallergenic acrylic glue. It is registered as a medical device of class Is with the Italian Ministry of Health.

Dermalize Pro Roll
Dermalize is available for professional tattoo artists in DERMALIZE PRO format (roll includes 10mt x 15cm), ideal for the first application after finishing the tattoo.
Dermalize Protective Tattoo Film is a latex-free breathable film, made of polyurethane and hypoallergenic acrylic glue. It is registered as a medical device of class I with the Italian Ministry of Health (Reg. 2366877/R e 2366878/R) , as well as with the American FDA (Food & Drugs Administration).
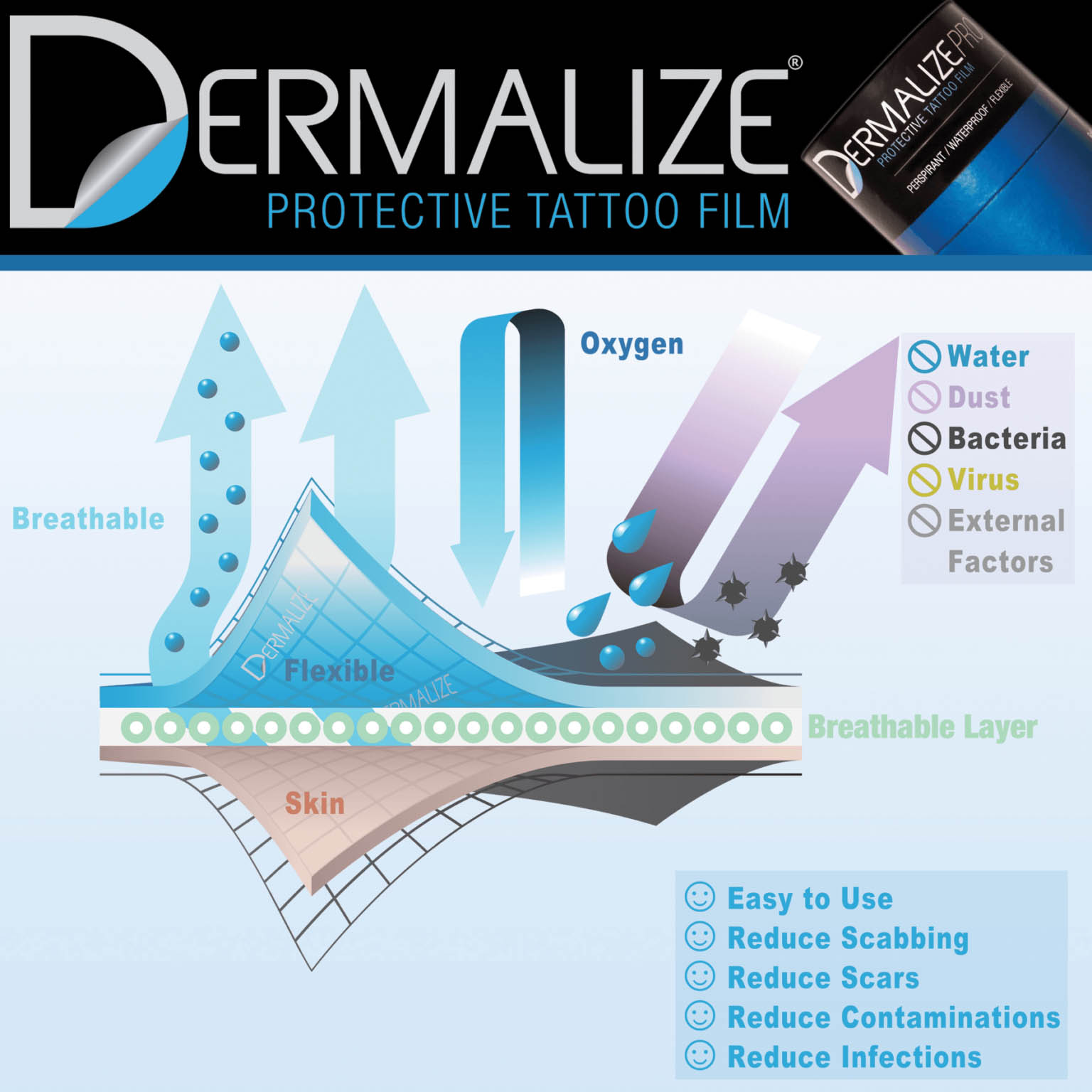
BREATHABLE
Dermalize (D-lizepro in the USA) sets itself on the global tattoo market as a certified medical replacement of the commonly used alimentary cling film. The film is breathable, letting oxygen in and allowing excess moisture vapor to be released from the wound.
ANTIBACTERIAL BARRIER
It builds an antibacterial barrier that guarantees a thorough protection from external agents. The tattoo film must be changed regularly as prescribed on the “How to use” section.
Any doubts left?
Check our FAQ’s section!
Where to buy dermalize





